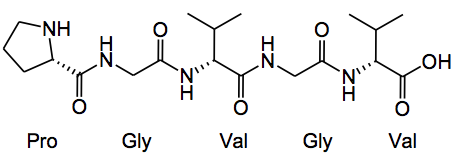
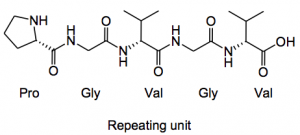
Here’s a new use for those gas tight Hamilton syringes we all have lying around in a lab gathering dust, as chemical reactors! This was utilised by Prof. K. Chiba and colleagues from the departments of applied biological chemistry and the department of chemical engineering, Tokyo University of Agriculture and Technology to prepare the repeating unit of elastin in a solution phase synthesis. So the target was to couple two repeating units:
Now this may not seem to be a great problem, however, amongst the physical properties of elastin are a hidden synthetic challenge: namely, the gelation and aggregation of the protein. This means that in solution, or indeed solid phase synthesis the whole lot clumps together and you can’t stir the soup. To overcome this Chiba has developed a hydrophobic benzyl alcohol (TAG) as a support allowing the use of less polar solvents for the solution phase coupling.
In spite of this “when coupling of Fmoc-Val-OH to H-Gly-Val-O- TAG was attempted in the biphasic solvent mixture using COMU and DIPEA, the reaction mixture became a highly viscous gel within 3 min and stopped the mechanical stirrer, indicating severe aggregation. The reaction was largely inhibited, and most of the starting H-Gly-Val-O- TAG was recovered. Although the addition of a large excess of cyclohexane and/or heating could also aid coupling, we expected that only reestablishing the fluidity of the reaction mixture was required since the substrate and reagents were both in “liquid” phase and their collision still potentially took place. Therefore, we connected two syringes to each other to fluidize the reaction mixture under high pressure“. So here are the Hamiltons:
The authors say that using the syringes as a low pressure reactor the reaction mixture is sufficiently fluid that it can be manually pumped from one syringe to the other. Of course, the reaction also speeds up and the conversion to product is consequently higher; no mention of any temperature increase. The reaction concentration is 0.1 M and 50 manual pushes are sufficient to provide grams of material.
The pressure required to do this is between 1 and 2 Bar. Now those of you who have used these gas tight syringes know just how hard it is to a) such anything up and b) push anything out. These post-docs must have thumbs of steel, 50 times at a pressure of 2 Bar! I sure hope this gets automated. The slightest blockage and you are screwed. Can’t go forward, can’t go back. And no mention of any temperature increases, this would worry me somewhat especially when your compounds are temperature sensitive. Assuming that you can get the RM from one syringe to another work-up is simple, “pour” it into acetonitrile and it precipitates as a filterable solid!!
I can just hear my pilot plant managers “you want to do what??”. That was a place where they did peptide synthesis every day. Still if it solves the problem of very viscous reaction mixtures then it is of use. So a useful tip for a not too uncommon problem.
![]()