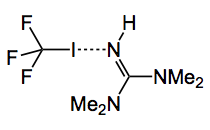
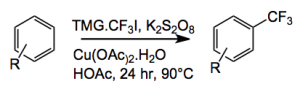Another trifluoromethylating agent was reported last week. It is a 1:1 adduct of trifluoromethyl iodide and tetramethylguanidine, TMG.CF3I, :-
It was discovered by the Ritter group at Harvard. It is easily formed and is a liquid at room temperature. This makes the dispensing of the trifluoromethylating agent easily done by syringe. The adduct is stable over several months. Other Lewis bases such as DMSO also form such adducts, although in this case 2 equivalents of DMSO are required.
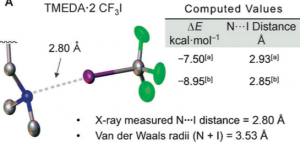
The dotted bond in the structure represents a halogen bond. Such bonding has apparently been known for more than 100 years and seemingly has application in supramolecular chemistry, but not much has been done by synthetic organic chemists. An x-ray structure of the TMEDA adduct was obtained, simply because the CF3 adduct was not crystalline:
also the former is not as stable as the latter. So what can you do with this compound?
Well you can use it to trifluoromethylate aromatic rings:
As usual, most functional groups are tolerated, well normal ones, ester, amide, nitrile. Steric congestion on the ring also does not matter. Average yields hover around the 50% mark. Aromatic heterocycles also trifluoromethylate well I say heterocycles, only one was examined, or at least presented in the paper, 4,6-dimethoxypyrimidine.
Throwing in a ruthenium catalyst and adding some light allows the trifluoromethylation of unactivated olefins. TMS enol ethers, and aldehydes required the use of the DMSO 2:1 adduct. It is not really obvious why: The authors suggest that “The ability of tetramethylguanidine to serve as one- electron reductant and/or H-atom donor..” may explain the differing reactivity.
And that’s it! No more examples.
There remains much work to be done investigating this compounds absolute generality, and it would have been nice to see a broader scope presented. Especially important would have been an indication as when to use the differing forms of the adduct. But it is a nice way of handling the otherwise gaseous trifluoromethyl iodide.
One gets an impression of speed when reading this paper. By that I mean, do the minimum as fast as possible to achieve acceptance. It could have been fleshed out a bit more to give us a better idea of the applicability of this reagent. But I’m sure that will follow after his move to the Max-Plank Institute at Mülheim in Germany.
![]()