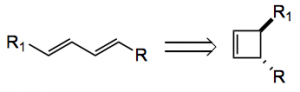
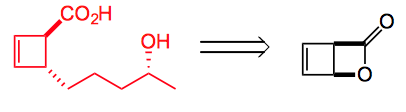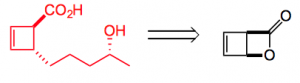As a method for the control of stereochemistry the use of carbocyclic systems has played a very large role in the success of organic synthesis. This week’s ASAP provides us with another example. The group of Maulide at the University of Vienna have employed stereodefined bis-cyclobutenes to stereoselectively prepare substituted 1,3- butadienes.
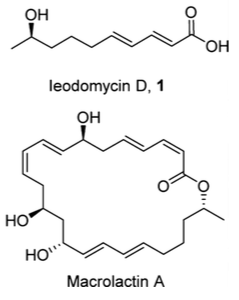
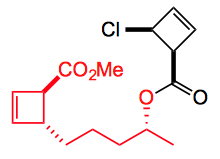
Now if you choose the correct R and R1 then you immediately recognise a substitution pattern present in many natural products. For example
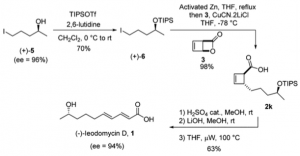
Let’s take the first compound leodomycin D. This requires the disubstituted cyclobutene which can be obtained from the β-lactone:
The lactone is easily obtained by photochemical cyclisation of α-pyrone. So forming the cuprate from an alkyl Grignard reagent in the presence of LiCl and exposing this to the lactone at -78°C gave the desired trans ring opened lactone in high yield and high dr. Even tBuMgCl reacts. This also works well with organozinc reagents which presumably undergo transmetallation to the copper species. So out comes a short asymmetric synthesis of (-)-leodomycin D:
Ring opening of the cyclobutene ring was achieved by thermolysis. So a 5 step synthesis in about 40% overall yield and 94 ee%.
This unit is present twice in macrolactin A. So simply making the bis cyclobutene
and following the same chemistry provides a fragment of the more complex natural product.
So here is a nice elegant approach to some complex natural products using “easy” chemistry to provide a valuable synthetic intermediate, a cyclobutene.Well done Vienna, I look forward to seeing many more synthesis employing this set of synthons.
![]()