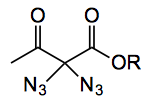
This week sees the disclosure of a “shockingly” neglected class of compounds, geminal diazides. Kirsch etal from the University of Wuppertal in Germany were the brave group synthesising these compounds. I will just add here that the word “shockingly” was the authors choice of terminology, however it is entirely appropriate when applied to geminal diazides! So the reaction is-
“the reaction of 1,3-dicarbonyls with iodine and sodium azide was typically performed in aqueous DMSO at room temperature: I2 (2.2 equiv), NaN3 (6.0 equiv), rt, DMSO/H2O (2:1, 0.1 M).”
Now they say 1,3-dicarbonyl compounds, their table is full of malonates, which are 1,3-dicarbonyls but there are other members of this class which were not reported. The yields are on average around 70% so it is worthwhile asking what is the rest of the material?.
There are a couple of slight inconsistencies I noted “Greatly to our surprise, however, the direct measurement of the accurate mass of several geminal diazides…. was rendered possible by standard ESI techniques. We reasoned that the geminal diazides derived from 1,3-dicarbonyls must be considered to be relatively stable.” This does quite not ring true when you examine the DSC data provided for three of these compounds. One of them, tert-butyl 2,2-diazidoacetate, has the comment on the DSC data (sup. mat.) …. sample decomposes during heating! The others measured produce significant amounts of energy upon decomposition, between 350 – 850 J/g. with onset temperatures around 140°C the lowest being 118°C. I think these onsets are on the low side for my liking. Given the rule of thumb that your process should be run about 80°C below the onset does not leave you much room for safe operation here. The authors did not add rust to the DSC samples, I’m guessing that the onset would decrease. To make matters worse the reaction is carried out with iodine and “We assume that, under our reaction conditions, iodine (or IN3) is the electrophilic iodine source that leads to the iodination of the easily enolizable 1,3-dicarbonyl system ….. Subsequent nucleophilic substitution gives the monoazide, which undergoes a second, probably more rapid, iodination followed by the second azide introduction. However, at this stage of our studies, we cannot rule out a radical mechanism”. So IN3 and a possible radical process. What could be better for your thermal safety than that combination?
Iodine azide has been known since 1900 where it was observed that “the solutions decompose quickly forming iodine and nitrogen, the crystalline residue left on evaporation of the ether, “would explode regularly and violently on touch- with a spatula“. There is actually a review of its chemistry. So I suggested my boss’s face would look amazed when I went to the plant with 100 kg sawdust, I imagine he would keel over in shock if I went with 100 kg of this reaction.
My azide was not explosive but on heating to the onset temperature a rapid pressure increase was observed (up to >6 Bar) which destroyed the thick stainless steel container. That was a mono azide, think about a diazide! So flow chemistry is the way to go here.
The authors do not provide any further chemistry that they may have done with these compounds nor do they make any suggestions. I’m left wondering what is a possible use, apart from fume cupboard destruction and employment for the fire and ambulance services? Thus is may be wiser to note the existence of this compound class and move on. I assume the chemistry department at Wuppertal is still standing.
![]()