Cycloadditions are a great way to build molecular complexity and if the partners contain heteroatoms it makes them an even better way. This week sees a paper from Wennekes (Wageningen University, Dreijenplein, The Netherlands) discussing functionalised nitrones and their cycloaddition reactions with alkenes.
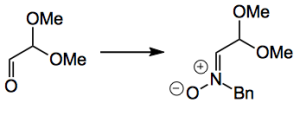
The nitrone used contained a protected aldehyde and is easily prepared-
“To a suspension of N-benzylhydroxylamine·HCl (10.0 g, 62.7 mmol) in MeOH (30 mL) was added NaOAc (6.68 g, 81.4 mmol, 1.3 equiv). The reaction mixture was cooled in an ice-bath and a 60% solution of 2,2-dimethoxyacetaldehyde in H2O (10.8 mL, 71.6 mmol, 1.13 equiv) was added dropwise over 5 min. The reaction mixture was stirred for 30 min. at 4 °C and then conc. in vacuo. To the residue was added DCM (500 mL) and H2O (100 mL) and the resulting layers were partitioned. The water-layer was extracted with DCM (100 mL, 2×) and the combined or- ganic layers were washed with sat. aq. NaHCO3 (100 mL, 2×) and brine (100 mL), dried (MgSO4) and conc. in vacuo. Affording 1 as a stable white solid (12.98 g, 99%). In our handling of the compound, nitrone 1 was stable for 1 year in the −20 °C freezer and for at least several weeks at room temperature”. So a nice easy method.
So as expected the [3+2] cycloaddition works well for a range of olefins with differing electronic and steric features. Nitriles and acetylenes also react well. All produce mixtures of syn and anti cycloadducts in varying ratios, some separable some not.
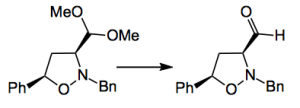
The whole point here is to liberate the aldehyde from the di-methyl acetal after the cycloaddition.
Guess what? Normal acetal hydrolysis conditions did not work! By normal I mean aqueous acid. They suggest the reason is the “…protonation of the nitrogen atom in the isoxazolidine ring in close proximity to the acetal might be suppressing methoxy protonation and thus the acidic hydrolysis.” After a screen TMSOTf/lutidine at -20°C provided the aldehyde in around 40% yield. What the rest of the material is, is not mentioned. So this is a reaction requiring some work. And it is probably better to move the aldehyde liberation to after the cleavage of the N-O bond. Indeed this is what happened.
Normal hydrogenation (Pd/C, H2) was slow and suffered from competing hydrogenolysis of the benzyl group. Raney nickel was effective and N-O cleavage was quantitative. Following this with a Pd/C catalyst gave the free amine which was protected. Normal acidic hydrolysis liberated the aldehyde in 70% yield-
Chirality can be introduced by using an olefin containing chiral groups (sugar olefins) or by employing a chiral hydroxylamine e.g. (S)-N-(α-methylbenzyl)-hydroxylamine.
So some useful chemistry here. Interesting is the acetal hydrolysis under basic conditions. The chemistry needs some work but higher yields are certainly possible.
![]()