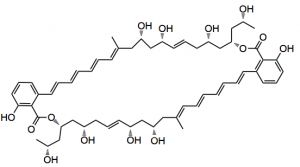
This week I spotted the asymmetric total synthesis of (+)-marinomycin, a potent anti-tumour/antibiotic, isolated from a marine actinomycete. It was presented by the Hatakeyama group from Nagasaki university. Now this is a large molecule a 44 membered macrodiolide which probably wont fit very well here but I will try:
You can see that the molecule is a C2-symmetric diester so an obvious route would be to synthesise the the mono ester and dimerise it in a double esterification reaction. This is a daring strategy as one could also expect the intra-molecular lactone to form as well as inter-molecular oligomers. So really it comes down to correct choice of protecting groups to assist the molecule in deciding what it wants to do.
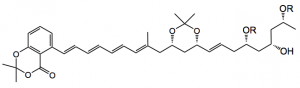
The authors chose a 1,3-dioxane protecting group as Evans had suggested that this provides a conformational benefit for macrolide formation (ring systems are always better for stereochemical control). This leads to the following compound as a di-esterificaion precursor:
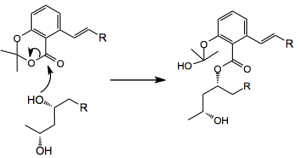
Considering the mechanism of the transesterification; it is expected to proceed as follows:
The di-esterification precursor could be retrosynthetically broken down into a Suzuki reaction, an acetylene coupling with an epoxide, inversion of a stereochemical centre and a Takai olefination.
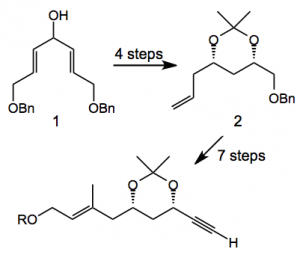
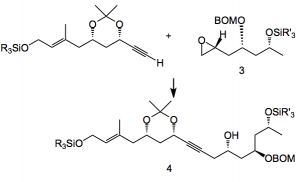
The route to the acetylene involved de-symmetrisation of the di-benzyl derivative 1 to give acetonide 2 in 4 steps, with an additional 7 steps to the acetylene in 36% overall yield:
Reagents: 1) Ti(OiPr)4 (10 mol%), d-DIPT (14 mol%), 4Å MS, TBHP, CH2Cl2, -25°C, 98% (99% ee); 2) DEAD, PPh3, p-nitrobenzoic acid, toluene, -30°C then K2CO3, MeOH, 74%; 3) Red-Al, toluene, 0°C to reflux, 82%; 4) 2,2-dimethoxypropane, PPTS, acetone, 99%; 5) PdCl2 (10 mol%), CuCl, O2, DMF/H2O (10:1), 82%; 6) (EtO)2P(O)CH2CO2Et, NaH, THF, 90% (E/Z=6:1); 7) DIBAL-H, CH2Cl2, -78°C, 99%; 8) TBDPSCl, imidazole, DMF; 9) Li, naphthalene, THF, -50°C, 96% (2 steps); 10) (COCl)2, DMSO, Et3N, CH2Cl2, -78°C; 11) MeCOC(N2)P(O)(OEt)2, K2CO3, MeOH, 88 % (2 steps).
Elegantly the required epoxide 3 for the coupling to the acetylene was also prepared from acetonide 2 in 7 steps and 51% yield. Coupling to 4 was achieved in 98% yield using the lithium acetylide and boron trifluoride diethyletherate.
Reduction of the acetylene to the (E)-alkene with Red-Al or LAH did not work and a workaround used a procedure devised by Trost. A few more manipulations and the authors arrived at the precursor for the transesterification.
Several conditions were examined, all based around the use of either potassium or sodium hexamethyldisilazide. The optimal set of conditions employed a dilute solution of the precursor (36 mmol) added to 4 x NaHMDS (14 mmol) in THF at -10°C over 30 minutes with further stirring at -10°C for 15 minutes. This delivered fully protected marinomycin in 71% yield with no recovered starting material. Global deprotection with perchloric acid gave (+)-marinomycin, 24 steps and 4% overall yield.
A nice synthesis, demonstrating that it is sometimes worth going for the direct method to obtain a particular compound. Some good chemistry in this paper and some elegant problem solving as take home messages.
![]()