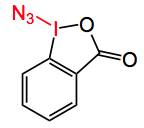
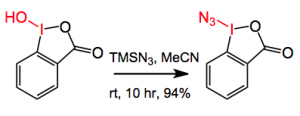
As we all know many compounds of biological relevance contain one or more C-N bonds. Occasionally the formation of these bonds can be a major headache. This weeks ASAP (which by the time of writing is no longer an ASAP!) is from the Hartwig group at Berkeley and appeared in Nature. In particular they were interested in forming C-N bonds at at tertiary alkyl group, which is particularly difficult at least under relatively mild conditions. The reagent used to achieve this transformation is the following iodoindane:-
This looks like a thermally challenged molecule prepared as expected by the exciting reaction:-
It melts with explosive decomposition at 138°C and is not shock sensitive.It adds the azide group to alkyl C-H bonds but usually this is done at higher temperature. This disadvantage makes it less suitable for the late introduction of a C-N bond in a complex molecule. Previous literature suggested that Fe and Mn-porphyrin complex could catalyse the reaction of sodium azide/iodosobenzene with hydrocarbons. So Hartwig surmised that the use of a different metal complex could be found that catalyses this reaction under milder conditions. Using cis-decalin as a model several different ligands were investigated, using 10 mol% Fe(OAc)2, 11 mol% of a ligand 2 equivalents of the azide regent in acetonitrile at rt, note the decalin isomerisation.
The yield was determined by further hydrogenation to the amine and carbamate formation, over the 3 steps, for the best ligand, was 75%, with a 4.3:1 mixture of diastereoisomers. The best ligand was:
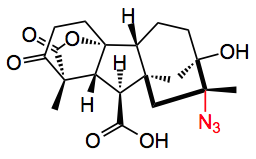
A series of hydrocarbons was then examined to demonstrate the selectivity for the tertiary carbon. The most impressive example was the synthesis of an azido tetrahydrogibberellic acid:
This was produced in 75% yield with 15% of the other diastereoisomer.
The mechanism was proposed to be a radical process, from the authors:”several observations reveal the general features of the mechanism. The site selectivities and stereochemical outcome of the azidation of cis- and trans-decalin and a-dihydropinene strongly suggest that a tertiary alkyl radical is generated. Attempts to use radical clocks to assess more directly a potential alkyl radical were hampered by the poor reactivity of the appropriate substrates, but the proposed radical intermediate is consistent with the selectivity for azidation of the more electron-rich, less polarized, and thus weaker, tertiary C–H bonds. Furthermore, addition of 1 equiv. of BHT and TEMPO, which are known to quench radicals, resulted in complete inhibition of the azidation reaction.“
So with the azide group in place one can imagine what sort of chemistry could come next. So a useful reagent here with nice mild tolerant reaction conditions. The only cloud is the thermal stability of the reagent especially in the presence of iron. This is something I would investigate before trying any reactions of a significant scale.
![]()