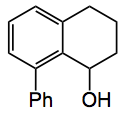
This week sees the birth of a new chiral auxiliary in Texas. The Montchamp group describe the synthesis of a (-)-8-phenylmenthol analogue which is cheap and readily available in both enantiomeric forms:
It is made, as one would expect, by a Suzuki coupling of the 8-iodotetralone which is obtained from the corresponding tetrahydro-1-naphthylamine. So the 5 steps produce the tetralone in about 35% yield. Straightforward chemistry here Sandmeyer included. Borohydride reduction gives the alcohol above in 100% yield.
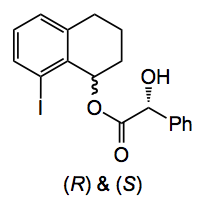
The authors went for chromatographic separation of the diastereoisomeric mandelic acid esters to get the enantiopure compounds. The 8-iodotetralone was also reduced and the mandelic acid esters separated by chromatography:
This compound can also be used directly in the Suzuki coupling and when followed by hydrolysis gives the chiral 8-phenyltetralol.
Now what is not clear is why they used chromatography to obtain the enantiomers. If, as they say, they wish to do this on large scale to make the auxiliary readily available then chromatography would not be my first choice. There are hosts of other methods out there. No mention of any of them in the paper, were they not investigated? Were they tried and didn’t work? We shall probably never know. This is a point that the referees should have picked up and at least a comment from the authors would have been welcome.
Anyway, does it actually work? Reaction of the pyruvate of the (S) alcohol with phenyl Grignard gave the addition product in 74% yield with a de of 96%.
(-)-8-phenylmenthol gave 92% de. Hydrolysis of the ester gave the enantiopure Grignard adduct and comparison with lit. values gives an ee of >95%. Here again this is the only example of its use as a chiral auxiliary! It would have been nice to complete this paper with a short reaction scope with various substrates and nucleophiles.
I know this is a communication; but really if you are going to propose a new chiral auxiliary a bit more in the paper would have been nice. Perhaps this will come, we shall see.
So nice work spoilt by lack of results which would have proved the usefulness, or not, of this compound.
![]()