I thought it would be good to suggest a publication of the week from amongst the ASAP material that I follow, a purely personal choice.
This week I picked the OPRD paper on the scale up of dimethyl cubane -1,4-dicarboxylate.
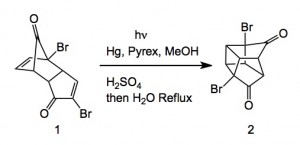
 The work appeared in Organic Process Research and development, 2013, doi.org/10.1021/op400181g . It was carried out by an Australian group, John Tsanaktsidis, Michael Falkiner, Stuart Littler, Kenneth McRae and Paul Savage from CSIRO and features a large-scale photochemical reaction which is very unusual to see in a scaled chemical process. Extending their previous work from 1997, they scaled the following reaction.
The work appeared in Organic Process Research and development, 2013, doi.org/10.1021/op400181g . It was carried out by an Australian group, John Tsanaktsidis, Michael Falkiner, Stuart Littler, Kenneth McRae and Paul Savage from CSIRO and features a large-scale photochemical reaction which is very unusual to see in a scaled chemical process. Extending their previous work from 1997, they scaled the following reaction.
As is the norm with such reactions the reaction requires high dilution to be successful. In this case they used a tailor made photochemical reactor. A solution of 1 in methanol/water was pumped through the reactor at 4 L/minute and the conversion of 1 to 2 was noted as 1g/4 minutes of irradiation. This meant a total time of 173 hours. Further processing of 2 through the double Favourskii ring contraction required significant development but eventually delivered the di-sodium salt corresponding to the di-ester of cubane.
One needs to be careful with these cubanes as they are, due to the highly strained nature of the system quite energetic materials, the do-acid and ester being more stable than the parent hydrocarbon. However the energy released upon warming above the melting point is not insignificant.
This paper represents, for me, a good demonstration of the scale-up of several very difficult chemical reactions, including excellent descriptive paragraphs of the problems and solutions. They are to be congratulated on a very nice piece of work.
![]()