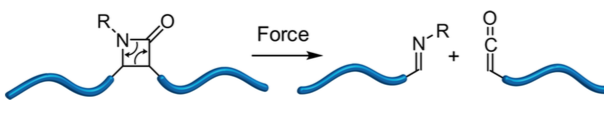
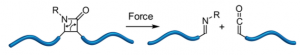
Staudinger may well have been pleased with this weeks ASAP. It deals with the use of polymers to pull molecules apart thus introducing us to the term mechanophore. This achievement comes from Moore at the Beckman Institute for Advanced Science and Technology and Department of Chemistry, University of Illinois at Urbana−Champaign, Urbana, Illinois.
It took a long time for chemists to synthesise β-lactams. One of the ways discovered was the use of a ketene/imine cycloaddition, which turns out to be very effective. Using mechanochemistry Moore and his group have done the reverse reaction.
By tugging on the ends of the blue polymer chains (polymers, another discovery by Staudinger [Nobel prize in 1953]) you can pull the 4-membered ring apart and re-form the imine and the ketene. This is a quite novel way of preparing these two synthons because “The ability to generate ketenes mechanochemically would have important implications for materials systems that are capable of responding to mechanical forces and repairing damage autonomically.”
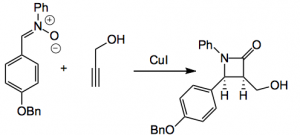
By carrying out some calculations Moore was able to estimate that the force needed to do this would be around 284 kJ/Mol or 3.47 nN, which even I might be able to exert. However the better way is to use sonication. The β-lactams were prepared by a copper catalysed Kinugusa reaction (a nitrone/acetylene cycloaddition) and produced the cis isomers.
A bit of fiddling around enabled this system to be incorporated in a polymethyl methacrylate polymer (PMA).
“All of the polymers in this study were synthesized with a number-average molecular weight (Mn) of 70−77 kDa and polydispersity index (PDI) of ≤1.09“. Control polymers containing the β-lactam unit at the end of the chain were also synthesised. Sonication for 2 hours resulted in a decrease in the MWt. of the polymer and the ketene generated was trapped by carrying out the reaction in isobutanol. Results were confirmed by NMR and UV spectroscopy. From the authors: “we have demonstrated that the β-lactam motif represents a new mechanophore capable of generating ketene and imine groups via a mechanically facilitated formal [2+2] cycloelimination reaction, the reverse transformation of the Staudinger cycloaddition. This finding is supported by comparisons of sonication-induced chain cleavage rates, UV− vis absorption measurements, and end-group analysis using 1H and 13C NMR spectroscopy. In addition, these experimental results validate predictions from DFT calculations (CoGEF). In view of the diverse reactivity of ketene groups and their versatility in polymer and materials science, the β-lactam mechanophore has outstanding potential for a variety of applications, including self-healing materials capable of autonomic restoration of mechanical damage”.
So self-healing materials. I have seen a couple of other papers on this topic recently. I suppose this means no more rips in the arse end of your pants, they will fix themselves within seconds just by creating some strain on the material, sonication preferred, saving serious embarrassment.
I also wonder if this polymer showed any anti-bacterial activity. Now that would be self healing.
![]()