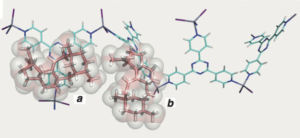
Once again the viability of the crystalline sponge x-ray methodology has been demonstrated as reported by Fujita. Following “genome mining of a terpene synthase gene from Emericella variecolor NBRC 32302 and its functional expres- sion in Aspergillus oryzae led to the production of the new sesterterpene hydrocarbon, astellifadiene (1), having a 6-8-6-5- fused ring system.“
So in combination with NMR the x-ray crystal structure of this new compound was determined to be
deduced from the x-ray picture of the terpene in the sponge:
There is just something about these pictures that makes me quite excited. To see the molecular structure of your compound is tremendous. One should note that “the X-ray Bijvoet method provides direct access to information about the chirality of molecules, but this compound exists as an oil and is not suitable for common diffraction studies. To the best of our knowledge, the crystalline sponge method is the only way to directly determine the absolute configuration of a compound without crystallization.”
So this was achieved with 10μg of the isolated product and the crystal complex was 150 × 100 × 50 μm3. So not what you would call large quantities or big crystals. To top this work off isotope incorporation experiments allowed a proposal of the biosynthetic pathway. Maybe some of those intermediates could also have been x-ray’d but perhaps this is the next step?
From the authors “In this study, the crystalline sponge method, coupled with an NMR study, readily established the absolute structure of a natural product immediately after its isolation. It should be emphasised that the combination of two reliable analysis methods completely eliminated any ambiguity in the other method (e.g., assumed conformation in NMR and heavy atom assignment in X-ray analysis).”
Hats off to crystalline sponges.
![]()