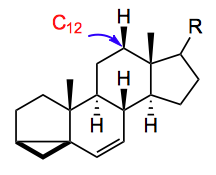
I see the Baran group is still investigating the oxidation of complex natural products. In this week’s ASAP they turn their attention to the steroid framework, examining oxidation at C-12.
After trying various “standard reactions”, with R being a methyl ketone or an alcohol, nothing promoted the selective C-12 oxidation. One very dry comment appears when mentioning a Hofmann-Löffler-Freytag reaction: “Attempts to generate a tethered radical were thwarted by the low reactivity of the C20 hydroxyl group, as we were unable to prepare the required carbamate for a ……“.
Fortunately help was at hand in the form of a paper by Schönecker which employed Cu complexes and molecular oxygen to biomimetically oxidise unactivated CH2 groups. So off they went to try and optimise the yield of this reaction which was reported to give a theoretical maximum of 50% yield and a practical one of 5%.
So using a pyridin-2-yl methyl imine and various sources of copper (mainly Cu(MeCN)4PF6) and reductants such as Zn, triethylsilane etc in acetone under an oxygen atmosphere between 23°C and 50°C for 90 minutes produced between 20 and 58% yield of the oxidation product. Changing the imine R group to 4-methylpyridin-2-yl)methyl, adding some methanol and using sodium ascorbate all at 50°C gave a 90% yield of the product!! So this is a 5 fold improvement, not to be sneezed at and to make things even better “…. the revised procedure is truly “dump-and-stir”, circumventing the laborious premixing, incubation, and complex workup required previously.“
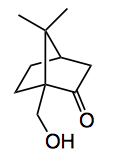
The reaction is applicable to a wide variety of steroidal substrates, even camphor giving a 94% yield of
compared to literature yields of between 4 and 31%.
NMR experiments were conducted to elucidate a possible mechanism of the reaction which is complicated and involves several Cu intermediates.
The utility of the oxidation was demonstrated by the synthesis of 3 important oxygenated pregnanes, pergularin, utendin and tomentogenin from which “Over 100 natural products with promising bioactivity can, in principle, be accessed from these three parent natural products, differing only in the location and identity of various ester and sugar side chains.”
So, as we have come to expect, from this group an extremely detailed publication and supplementary material. It again highlights the necessity of “keep on trying” and mass balance, the latter being important for possible mechanistic insights and unexpected new compounds.
![]()