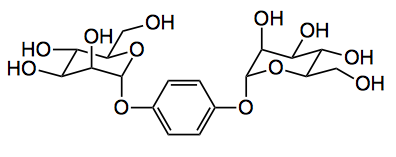
Fujita gave us crystalline sponges for x-ray analysis of difficult to crystallise compounds now he presents us with a sugar sponge! In his latest development he employs a mannose based host the basic element of which is:
“When this ligand was slowly combined with NaOH in EtOH/water/Et2O by a layer diffusion method, colourless needle-like crystals formed on glass wall. After washing the crystals with Et2O, sugar sponge [(ligand)2(NaOH)2. (Et2O)(H2O)]n was obtained in 65% yield“.
The sugar sponge selectively accommodates hydrophilic guests in diisopropyl ether and enables X-ray diffraction analysis of them. So after soaking the sugar sponge in a mixture of diispropylether and n-propanol only n-propanol is incorporated into the channels allowing X-ray structure determination of n-propanol! Interestingly “In the structure of inclusion crystal [(ligand)2(NaOH)2. (Et2O)(H2O)]n.n-propanol, two crystallographically unequivalent guests (guest A and B) were found with 100% occupancy”. This is presumably due to various hydrogen bonds existing within the host.
Similar experiments were done using (S)-propylene oxide providing wonderful pictures:
I always knew this was the correct structure!
In the words of the author “Since sugar sponge is composed of both hydrophilic and hydrophobic parts, not only hydrophobic but also hydrophilic guests can be analyzed by the crystalline sponge method. Although the target compounds are rather limited to small molecules because of its narrow-sized channels, tight and strong binding of guest molecules are quite important to obtain X-ray data at the good quality.” and “We believe that sugar sponge 2 has expanded the scope of the crystalline sponge method by covering a shortcoming of the prototype crystalline sponges.”
![]()