Here is a nice synthesis of (+)-chloriolide from Ostermeier and Schobert from the University of Bayreuth in Germany which appeared in this weeks JOC ASAPs.
This simple looking macrolide can be isolated from a fungus found on decaying wood and did not possess any antibiotic activity. I said simple looking, however previous routes were relatively long (around 20 steps) with low overall yield. This synthesis starts from two cheap readily available enantiomerically pure compounds; lactic acid and cellulose.
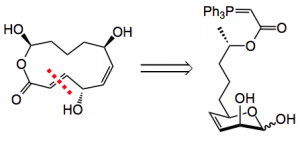
The retrosynthetic analysis shows an interesting disconnection, a E selective Wittig reaction was chosen as the ring closing step:
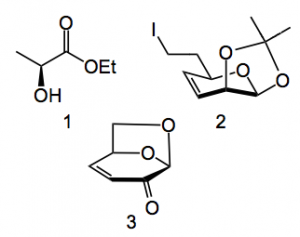
This ring closure has been previously investigated by this group and found to be viable for macrocodes of ring sizes 12 to 18 however it was unclear if the aldehyde would be present in sufficient amounts at the high pH required for the Wittig. Working through the retrosynthetic analysis lead to ethyl lactate 1 and compounds 2 & 3.
Ketone 3 was obtained in 2% yield from the pyrolysis of cellulose at 500°C! This is apparently easy to carry out in the lab on a large scale. Ten steps delivered the iodide 2 using conventional chemistry. If one ignores the 2% yield the sequence is quite efficient. and 3 is obtained in around 20% yield. Installation of the ylide was straightforward in an eight step high yielding sequence producing ylide 4:
The ylide underwent immediate Wittig cyclisation delivering the desired compound in 65% yield.
The pyrolysis of cellulose is an unusual way of obtaining carbohydrates with interesting structures and I have not seen this applied in synthesis before. It’s a great use for all the surplus paper produced by big pharma and other large institutions. This is a very interesting publication with lots of useful chemistry and I can recommend it as a good read.
![]()