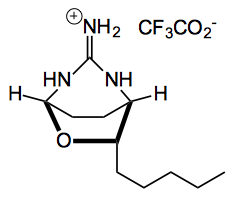
This week saw the synthesis of the alkaloid (+)-monanchorin. Isolated from a sponge it showed promising cytotoxic activity.
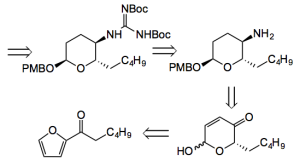
O’Doherty and Ma from Northeastern University, Boston used a Noyori reduction of an acylfuran to introduce asymmetry. Interestingly this route also relied upon an Achmatowicz rearrangement, a diastereoselective palladium catalysed glycosylation, reductive amination, and an acid catalysed bicyclic guanidine mixed acetal formation. All this in only 9 steps! Their retro-synthesis goes-
The acyl furan is easily obtained and reduced in high ee using Noyori’s catalyst to the alcohol, ready for the rearrangement.
They chose the Achmatowicz rearrangement because from previous work it was recognised that this reaction was useful in the synthesis of pyrans and they observed the stereochemistry of the alkaloid pyran ring is that of amicetose (another new sugar for me).
A 2-step Pd catalysed procedure was used to diastereoselectively protect the anomeric OH [(Boc2)O, DMAP then PMBOH, Pd(PPh3)2 ].
The amine was synthesised by a procedure involving stereoselective carbonyl reduction followed by mixed carbonate formation with methyl chloroformate then Pd catalysed displacement with azide and hydrogenolysis –
Interesting here is “Exposing carbonate to the conditions developed by Sinou (TMSN3, (Pd(ally)Cl)2/1,4-bis(diphenylphosphino)-butane) afforded a single regio- and stereoisomeric allylic azide in a good yield (70%)”.
Further reaction of the amine with N,N-bis-Boc-S-methylisothiourea in the presence of silver nitrate and triethylamine resulted in a bis-Boc protected guanidine . “Treatment of which with TFA in a one- pot reaction removed both Boc-protecting groups and induced the final ring closure furnishing (+)-monanchorin (1) in excellent yield (98%)”.
A quick look at the supplementary info. for this last reaction shows it was done on a very small scale, 0.122 mmol, and purified by flash chromatography on silica-gel. Isolated was the TFA salt of the alkaloid as an oil! The chromatography solvent was 5% DCM/IPA but the Rf is 0.08 in 5% DCM/MeOH). Might have helped to have the Rf in the chromatography solvent!
So a nice route to quite a complex little molecule in 9 steps and 22% overall yield. Nice paper and easy reading. The scale could have been larger but I suppose it is research!
![]()